Alkalıne Batterıes
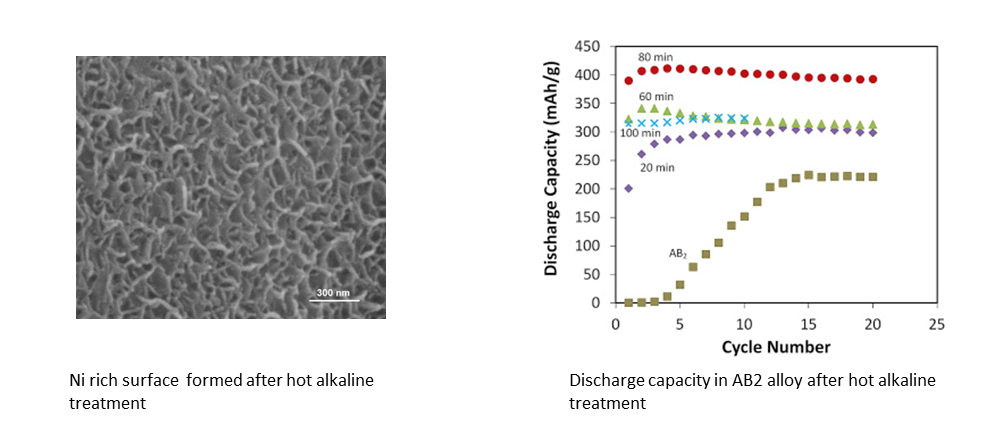
Efforts to improve alkaline batteries concentrate on both anode and cathode materials. Materials under active research are listed below. In terms of anode material, greater portion of effort have concentrated on the development of rare-earth free metal hydrides . Considerable progress has been made with regard to AB2 mult-component alloy alloys where a capacity in excess of 400 mAh/g could be obtained without much difficulty in activation.
| anode |
| AB2 |
| Mg |
Mg and Mg based alloys offer storage capacities much higher than those achieved with AB5 or AB2 compounds. Unfortunately hydrides of these alloys are too stable for reversable reaction. Currently Mg50Ni50 alloy have suitable thermodynamics, but other compositions could well be developed as suitable anode materials . Unfırtunately Mg and Mg based alloys are not durable in alkaline envorionment and therefore are subject to fast capacity decay. In a recent work we have successfully wrapped Mg50Ni50 electrode into a nafion envelope . This has improved capacity retention, and suprisingly has led to an increase in the storage capacity of the alloy. The coating would adversely affect the charge transfer resistance.
Since the design of NiMH batteries is dictated by positive electrode, there is much interest in improving the discharge capacity of Ni(OH)2 positive electrode. Forms of Ni(OH)2 with much higher capacity is available but with a low tab density making electrode preparation rather difficult. Currently work is in progresss which considers electrode architecture with improved gravimtric and volumetric energy density. Efforts to replace Co additives with lower cost alternatives is also in progress.
| Cathode |
| Ni(OH)2 |
| Mn(OH)2 |
Zn-Mn are low cost primary alkaline battery with a potential that could be made rechargeable. The positive electrode MnO2 with a form delta-MnO2 could reversably be reduced to Mn(OH)2 with two electron exchange. Currently work is in progress based on combinatorial material chemistry to identify compositions that would help improve the reversability with an improved discharge potential profile.